If you were to zoom in on your body—past the skin, past the cells, right down to the molecular “basement”—you wouldn’t find a soup of random chemicals. You would find an engineering masterpiece.
To understand how our DNA Goes Public 3D models work (and why they physically transform into RNA), we first have to understand the three “building blocks” that make life possible. If you’re a homeschooler or a student, think of this as the “User Manual” for your own existence.
The Nucleotide: Life’s Data Unit
Think of a Nucleotide as a single Lego brick that also happens to be a USB drive. It carries structure and information.
Every single nucleotide is made of three rigid parts locked together:
-
The Phosphate Group (The Glue): This is the tough connector. It hooks onto the sugar of the next unit to create the “backbone” of the DNA strand.
-
The Sugar (The Scaffold): In DNA, the sugar component is deoxyribose, which differs from ribose by lacking an oxygen atom at the 2′ position of the sugar ring (the 2′ hydroxyl group is absent); in RNA, it’s Ribose. It’s the central hub that holds everything in place.
-
The Nitrogenous Base (The Code): This is the variable part—Adenine, Thymine, Cytosine, or Guanine. While the “glue” and the “scaffold” stay the same, the base changes. The order of these bases is the literal digital code that tells your body how to build you.
Pro-Tip: If you have the Sugar and the Base, but you haven’t added the Phosphate “glue” yet, scientists call it a Nucleoside. Add the Phosphate, and it becomes a Nucleotide.
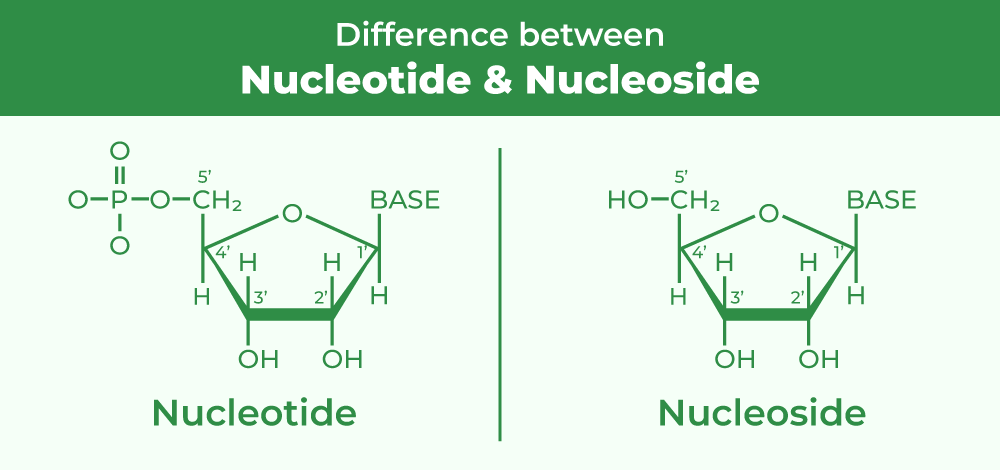
Image source: Geeks for Geeks
Amino Acids: The Raw Materials
If DNA is the blueprint, Amino Acids are the lumber, bricks, and steel. They link together to form Proteins, which do the heavy lifting: building your muscles, fighting viruses (antibodies), and speeding up chemical reactions (enzymes).
Every amino acid has a “Universal Plug” (the Amine and Carboxyl groups) so they can all snap together in a line. But they also have an R-Group (The Side Chain). This is the “Special Feature.”
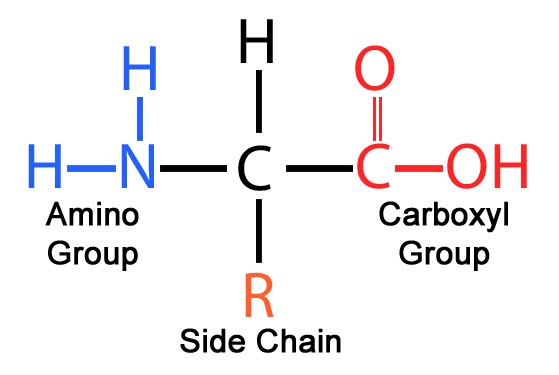
Some R-groups love water; others hate it. Because of these different characteristcs, the long chain of amino acids starts to fold, twist, and snap into a 3D shape.
The R represents the chemical group that distinguishes each amino acid from the others and gives them a definite name. Each radical group has a different size (number of atoms) and physicochemical characteristics that become properties of each amino acid and determine the strength and the nature of both intra- and inter- molecular interactions.
In biology, shape is everything!
The “Bio-Signature”: Why Life Isn’t Random
This is where it gets fascinating. If you look at amino acids found on a meteorite or created in a random lab experiment, they look messy. But inside a living cell, they are mathematically precise.
There are two massive differences between “random” chemistry and “living” chemistry:
The “Left-Hand” Problem (Homochirality)
Most molecules have a “twin.” They come in Left-handed (L-form) and Right-handed (D-form) versions. They are mirror images, like your own hands.
-
In the Wild: Nature usually produces a 50/50 mix (a Racemic Mixture).
-
In Life: Your body uses 100% Left-handed amino acids.
Why does this matter? Think of building a structure with Lego bricks where every connector is designed to fit in one orientation. If you insert a brick with reversed connectors, the structure won’t lock together and collapses. Life requires 100% “Handedness” purity to build stable, working 3D proteins.
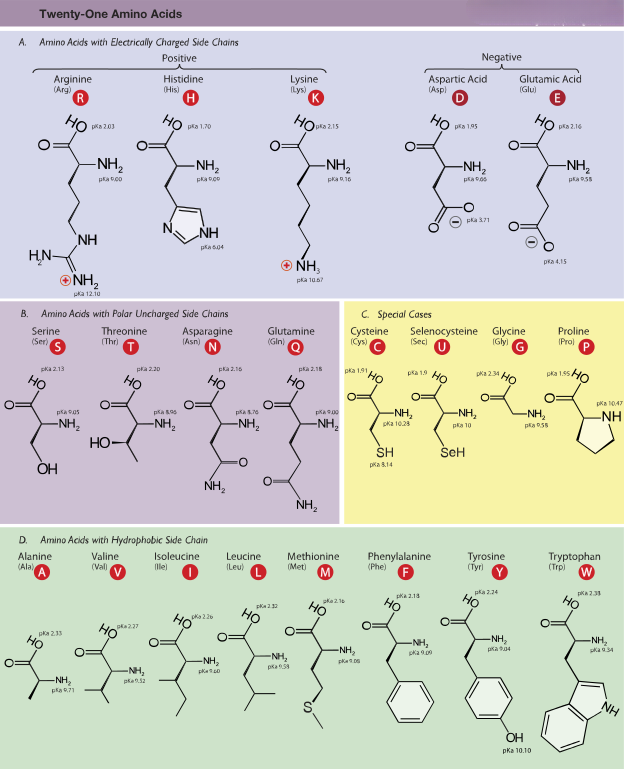
The “Magic 20” Alphabet
There are hundreds of types of amino acids in the universe. However, every living thing on Earth—from a blade of grass to a Great White Shark— by far the most important are the 22 α-amino acids incorporated into proteins.
Life “hand-picked” a specific alphabet of about 20 units that are perfect for folding into complex machines. It’s not a random collection; it’s a curated toolkit.

Why the 3D Shape Matters
You can read about DNA in a textbook, but you can’t truly feel the “logic” of the code until you see how these pieces fit together in space. That is why we created our dynamic 3D Model—to turn these invisible chemical “hands” into something you can hold.
Ready to see how these nucleotides snap together to form the famous Double Helix?
Check our store and feel free to get a free perk from our website today!